A Kinetic and Mechanistic Study of Palladium (II) Catalysed Oxidation of Arginine by Cerium (IV) in Acid Media

Redox trends in cyclometalated palladium( ii ) complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT03786K

Redox trends in cyclometalated palladium( ii ) complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT03786K

Redox trends in cyclometalated palladium( ii ) complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT03786K

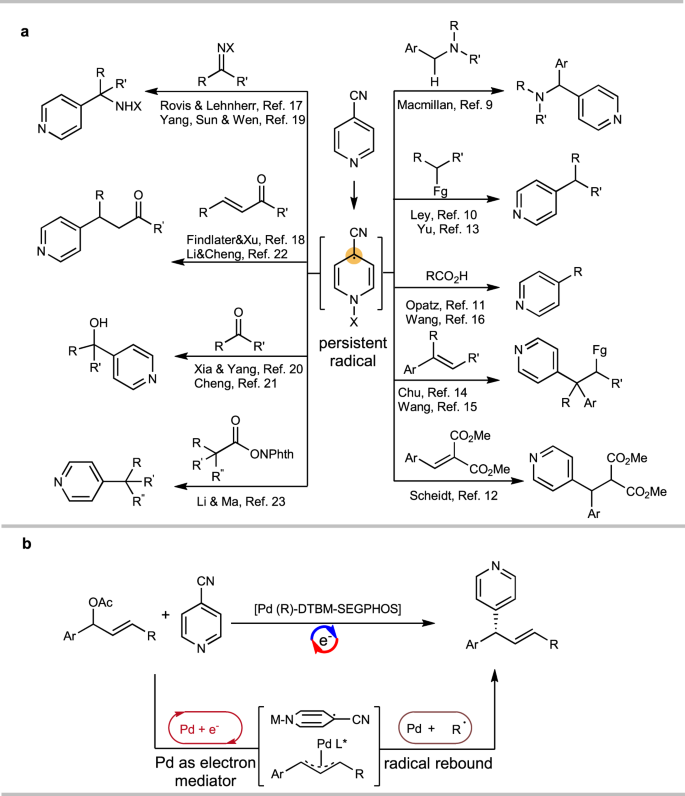
Electrochemical Methods for Pd‐catalyzed C−H Functionalization - Erchinger - 2021 - Asian Journal of Organic Chemistry - Wiley Online Library

Can Donor Ligands Make Pd(OAc)2 a Stronger Oxidant? Access to Elusive Palladium(II) Reduction Potentials and Effects of Ancillary Ligands via Palladium(II)/Hydroquinone Redox Equilibria | Journal of the American Chemical Society

Enhanced Electrocatalytic Reduction of Oxygen at Electrodes Coated with a Multi-Metallic Co(II)/Pt(II) Porphyrin

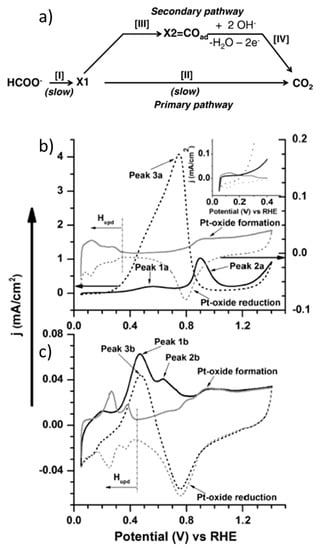
Electrochemical behavior of CO2 reduction on palladium nanoparticles: Dependence of adsorbed CO on electrode potential - ScienceDirect

Bio-inspired lanthanum-ortho-quinone catalysis for aerobic alcohol oxidation: semi-quinone anionic radical as redox ligand | Nature Communications

Palladium Oxidase Catalysis: Selective Oxidation of Organic Chemicals by Direct Dioxygen‐Coupled Turnover - Stahl - 2004 - Angewandte Chemie International Edition - Wiley Online Library

Accurate Oxidation Potentials of 40 Benzene and Biphenyl Derivatives with Heteroatom Substituents | The Journal of Organic Chemistry
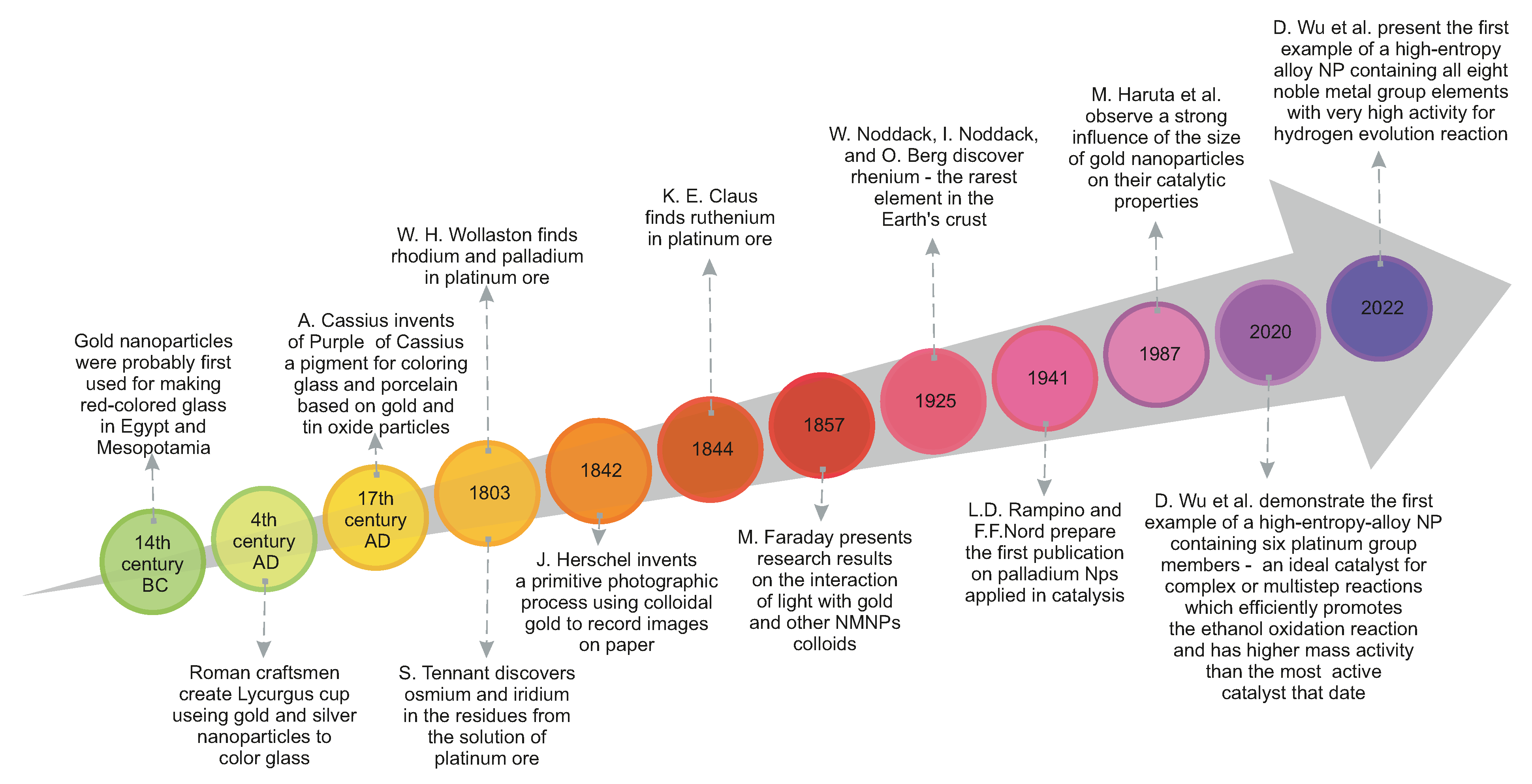
Crystals | Free Full-Text | Carbon-Supported Noble-Metal Nanoparticles for Catalytic Applications—A Review
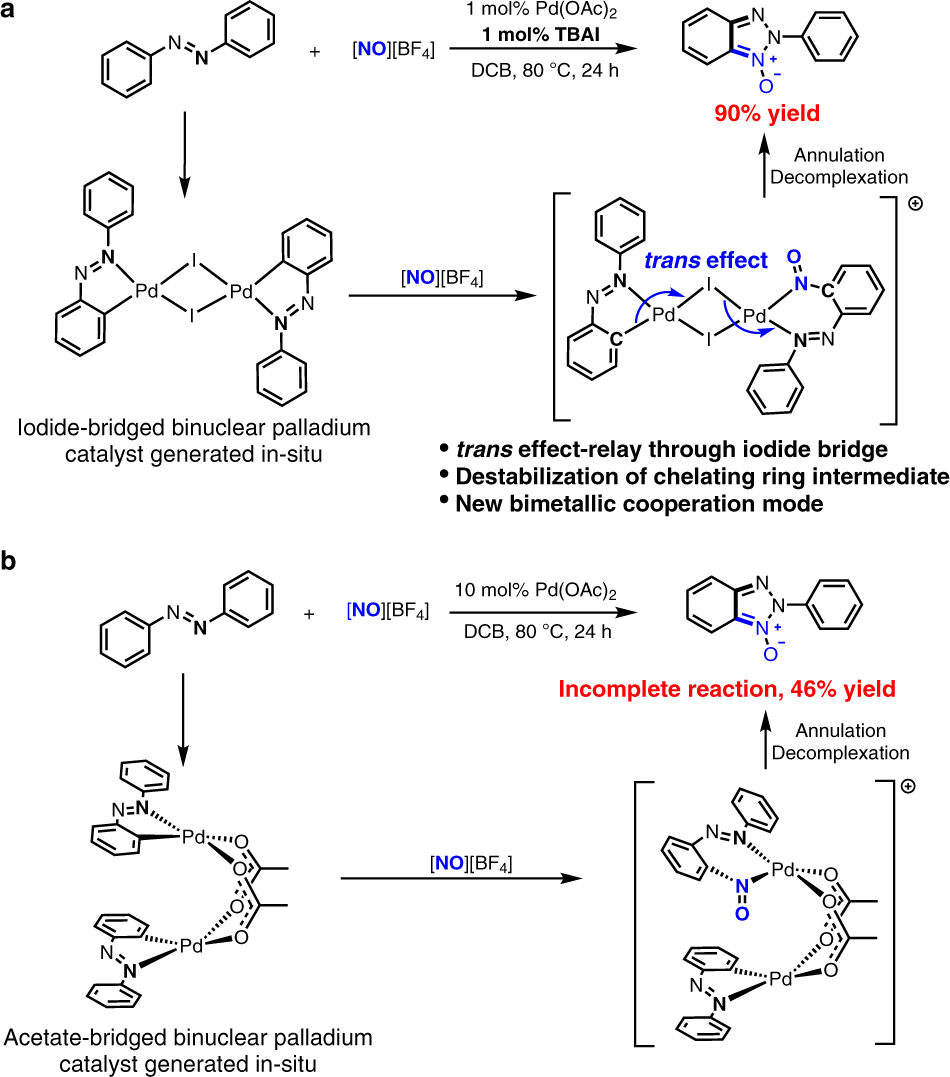
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry

Electrochemical behavior of CO2 reduction on palladium nanoparticles: Dependence of adsorbed CO on electrode potential - ScienceDirect

Can Donor Ligands Make Pd(OAc)2 a Stronger Oxidant? Access to Elusive Palladium(II) Reduction Potentials and Effects of Ancillary Ligands via Palladium(II)/Hydroquinone Redox Equilibria | Journal of the American Chemical Society

The oxygen reduction reaction on palladium with low metal loadings: The effects of chlorides on the stability and activity towards hydrogen peroxide - ScienceDirect